Lemon Battery Experiment Explanation:- Hello, my dear students welcome to my blog PogoBoss.com I am Rishabh Mishra founder and CEO of this blog website, today in this blog post I am going to show a science experiment on “How to Make a Lemon Battery“, two different metals in the form of zinc and a copper nail are inserted into a juicy lemon, so please read all the steps carefully.
Aim- Can we produce electricity from lemon? | Lemon Battery Experiment
Producing Electricity–
Vinit, you know, torch uses the battery for the glow.
Yeh! But I don’t know how the battery produces electricity.
Don’t worry. I’ll see you how the battery produces electricity.
Let’s see, how can we produce electricity with the lemon battery,

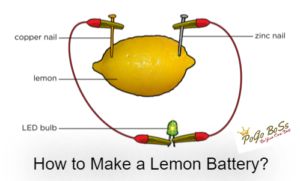
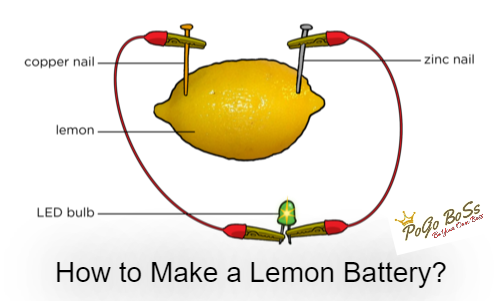
Things Needed
• One LED bulb
• Two electric wires (about 30 cm in length)
• One lemon
• Two long thin objects (made of different metals such as steel paperclip and copper-nail)
Now follow these steps:
Step-1: Gently squeeze the whole lemon to make it juicer inside.
Step-2: Poke the two different nails of metals or graphite electrodes into the lemon.
Step-3: Attach the wires to the electrodes and also to the LED bulb to make a circuit.
What happens now
You will observe that the LED bulb glows.
How it happened
The lemon works like a battery and produces electricity. An electric cell is made from two electrodes of different types (different metals or graphites) and an electrolyte solution.
Conclusion
The acid in the lemon and the two different metals create the electricity that flows through the wires to an LED bulb.
Hope you have successfully completed the How to Make a Lemon Battery science experiment, if you have any doubts regarding this science project you can ask below in the comment section. For more content like this, please visit here:- Blog
Also Read- Moral story on Forgive and Forget in English
Please share this post with your classmates from the share button that is present below this.
Thanks
Have A Nice Day!


1 Comment
[…] How to Make a Lemon Battery? […]